Presentation
This session presents a high temperature solid oxide fuel cell model, SOFC, burning pure hydrogen. This two parameter model is quite simple. We will show how to establish the balances of the species and the energies put into play.
Two other sessions allow to refine this first model.
In session S62En, we will take into account the polarization equation of the cell, and then the cooling of the cell. Lastly, an exercise will show how the cell can be coupled to a gas turbine pour form a high efficiency cogeneration installation.
In session S63En, we will explain how to modify the previous models in order to replace hydrogen by a fuel such as methane.
(Session realized on 09/14/11 by Renaud Gicquel)SOFC CELL
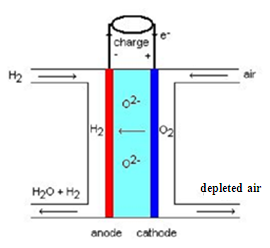
- 600-1000°C
- water at the anode
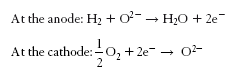

SOFC tubular cell
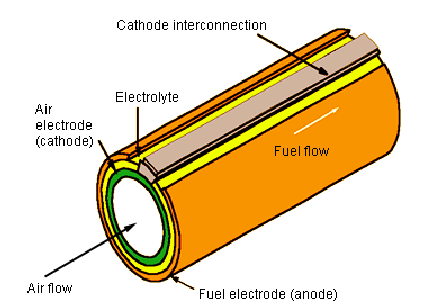
- cylindrical technology
- Φ = 19 mm
- L = 1.5 m
Doc. Siemens Westinghouse
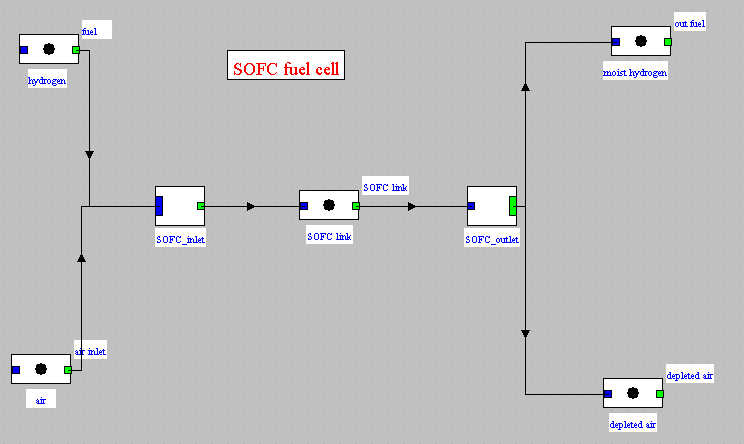
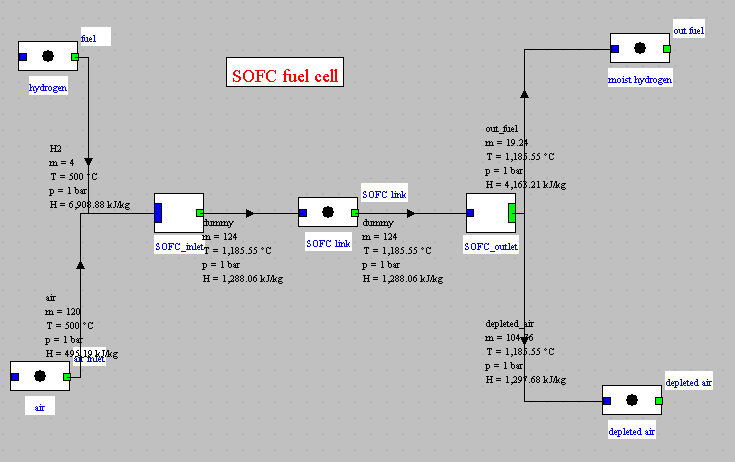
Thermoptim diagram
Balance of species
- τ : rate of hydrogen use
- anode reaction ( kJ/kmol)
- at the anode outlet:
- λ: air factor
- at the cathode inlet:
- at the cathode outlet:
Energy balance
- Rate of fuel use τ
- Energy efficiency ε
- Electric power:
- Thermal power:
First model
We will now show the results provided by the two-parameter model, and how it is implemented as an external class.
This exercise is explained in eight steps, grouped into two main parts:
- The first three show the results provided by Thermoptim
- The other five explain how is made the computer implementation of the external class
This model uses special Thermoptim components called external classes, that extend its core library, and whose use is explained in a note downloadable from the links below.
The study of the second part is reserved for those with sufficient prior knowledge of the mechanism of external classes. If this is not your case, you can either refer to the session S07En_ext, which will allow you to get started with it, or skip this part by going directly to the final step.
All work files corresponding to this model can be downloaded from the links below called First model files.
Westinghouse 100 kWe cell
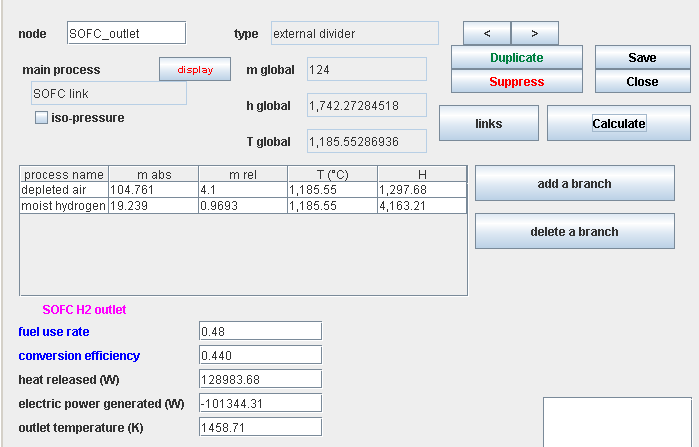
Synoptic view of the cell
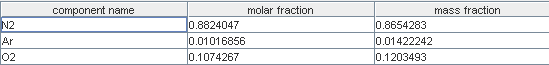
Composition of gases
- humidified hydrogen , flow 19.24 g/s LHV: 11 230 kJ/kg
- air depleted in O2, flow 104.76 g/s

Model implementation
The next steps explain how the model is implemented in the external class.
First, to ensure consistency of the model (to avoid that the inlet mixer be connected to an inadequate outlet divider), each of the two nodes tries to instantiate the other by seeking its class among the project external components and verifies that both are connected to the same link process. If the operation fails, a message warns the user that the construction is incorrect. This check is performed by the methods setupInletSOFC() and setupOutletSOFC().
In addition, tests of consistency of each node are carried out by the method checkConsistency() to check the fluids are well connected: in this case, air and hydrogen as input, and two outlet gases, the identification of the depleted air being done on the basis of the presence of nitrogen.
Please refer to Volume 3 of the reference manual for explanations on this point, valid for all external nodes.
The study of the method calculateProcess() of the external class SOFCH2Outlet shows how the model has been implemented
As explained in the next steps, calculations are made in six very simple steps.
Study of the external class (1-2)
- 1) the parameter values are read on the screen
- 2) the inlet molar flows are provided by the inlet mixer


Study of the external class (3-4)
- 3) the anode outlet molar flow and composition are determined
- 4) the cathode outlet molar flow and composition are calculated
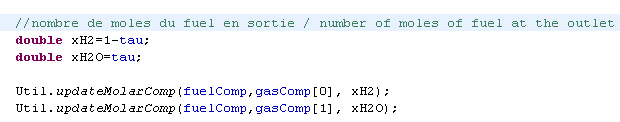

Study of the external class (5)
- 5) the mass flows are calculated and updated

Study of the external class (6)
- 6) the energy calculations are performed and the outlet temperature determined



Extensions and sensitivity studies
With this model, which is presented in Part V of the book Energy Systems, several sensitivity studies and extension activities can be considered:
- First, change the values of hydrogen and inlet air flows as well as τ and ε, to see how changes the stack performance. You will note in particular that, for a given flow of hydrogen, it is necessary that the air flow exceeds a threshold value below which the mole fraction of oxygen at the cathode outlet becomes negative (there is not enough oxygen available in light of hydrogen available). This is a failure of our model, which should alert the user in such cases
- Analyze how the model is built, and possibly refine it by adding, for example an electric model to calculate τ and ε according to the intensity required
- Modify the model to consider the case where the stack is cooled by an external fluid (addition of a thermocoupler)
- Insert the stack into a more complex system coupling it to a gas turbine
- Change the model so that the stack accepts a fuel other than pure hydrogen (e.g. methane)
These activities will be presented in the sessions S62En and S63En.