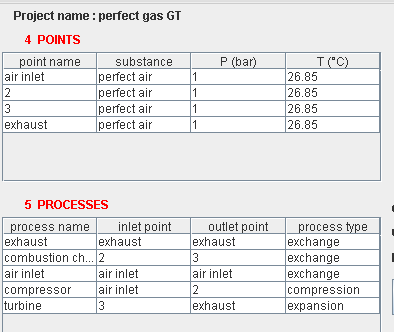
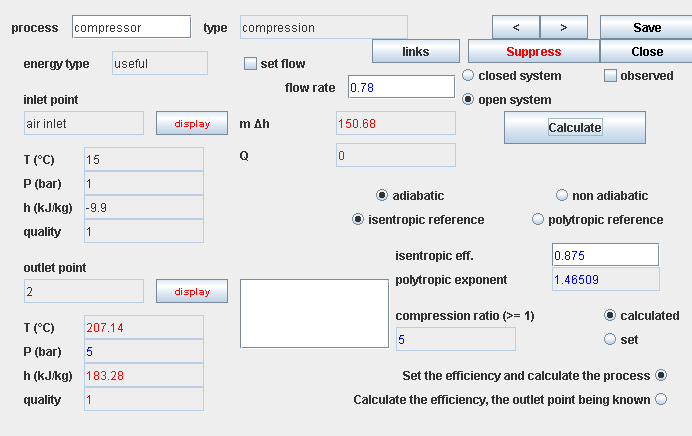
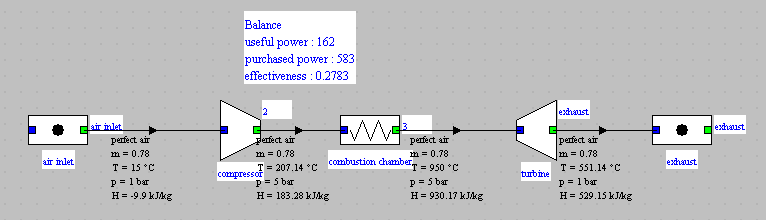
Presentation of the exercise
This exercise is primarily intended to illustrate how to calculate adiabatic compressions and expansions. It is centered around the study of a small capacity gas turbine, called micro-turbine. This micro-turbine is an existing machine, used for several applications, including cogeneration.
In session S46En we will give a cogeneration facility example based on this machine.
This exercise is divided into three parts:
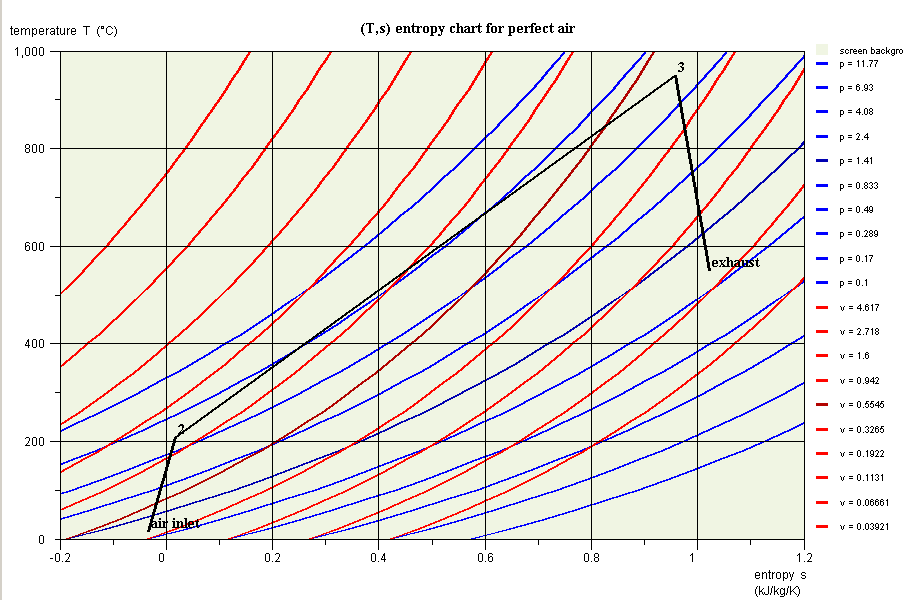
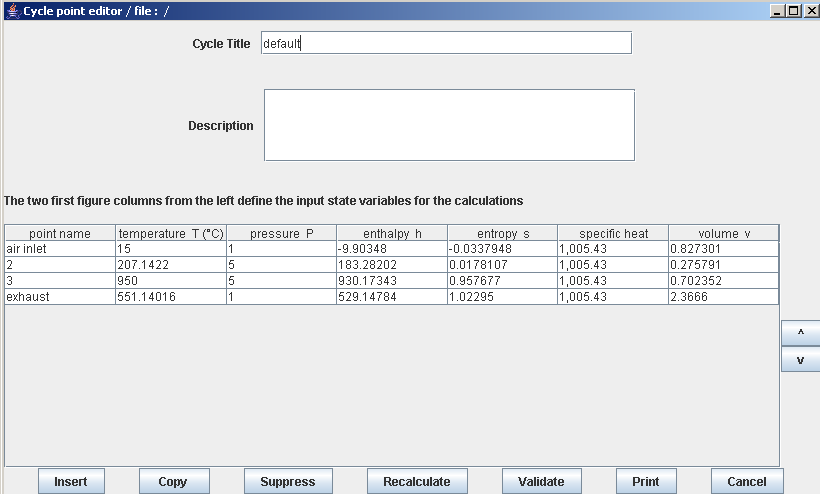
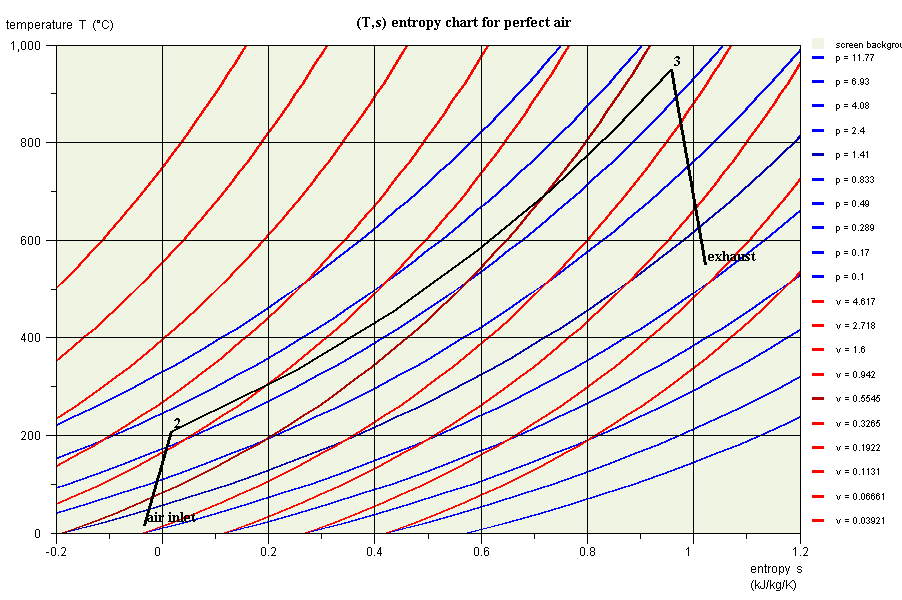
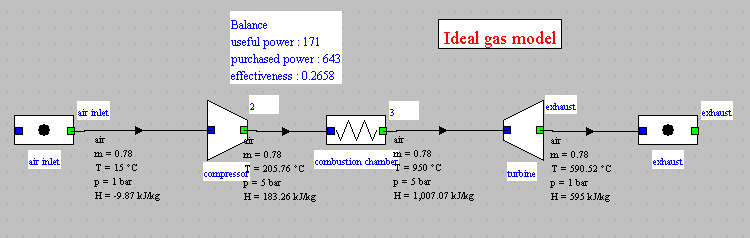
- in this part (session S21En) we assume that the machine is traversed by a single gas, air, initially modeled as a perfect gas, and in a second time as an ideal gas. The resulting model is obviously simplistic, but it is interesting for beginners, because it allows them to make the connection between analytical theoretical calculations and resolution in Thermoptim.
- in the second part (session S22En), we consider the combustion reaction that takes place in industrial machines. The model is much more realistic. The comparison with the analytical model allows us to analyze its limits
- in the third part(session S23En), we analyze quantitatively the irreversibilities of the cycle by setting up its exergy balance, and deduce ways of improving the machine cycle (regeneration).
Before starting working with Thermoptim, we strongly recommend that you study session S07En_init, which introduces all the concepts which you will be using.
(Session realized on 06/16/11 by Renaud Gicquel)