Exergy balances
This session explains how to set up the exergy balance of a cycle.
The theory of exergy (the link below gives you access to the relevant textbook excerpt) provides a quite rigorous framework to quantify the thermodynamic quality of any system, open or closed, in steady-state or not.
It is increasingly accepted as the preferred tool to compare and optimize thermodynamic cycles, thanks to exergy balances.
It defines an integrated analysis method that includes the first two laws of thermodynamics, and thus allows us to take into account both the amount of energy put into play and its quality, which the first law cannot do.
Course reference:
- “Fundamentals of thermodynamics / Exergy”
- “Cycles / exergy balances”
To follow the presentation, go to the next step
(Session realized on 06/16/11 by Renaud Gicquel)ENERGY BALANCES
- First law
- do not take into account the quality of energy:
- 1 kWh at 1000 °C = 1 kWh at 20 °C
- Second law:
- exergy: function xh = h - T0s
Exergy, exergy balances
- exergy depends on both the system state and the chemical composition of its elements
- exergy is destroyed whenever irreversibilities exist
- main interest of the exergy balances: study the distribution of irreversibilities in a system
Heat-exergy
- Δxhi = T0 Δsi ≥ 0 exergy dissipation (losses)
heat-exergy received from source at temperature
Maximum engine work
- maximum engine work provided by an open system: sum of exergy-heats of the sources, less change in exergy of the fluid passing through it and exergy destroyed because of irreversibilities
Calculation of incoming and outgoing exergies (1/2)
- composed of four terms:
- physical, kinetic, chemical and potential exergy
- kinetic and potential exergy are equal to the energies of the same name, generally negligible for energy systems that we study
- physical exergy: function xh = h - T0s
- evaluated in reference to the conditions of T and P of the environment
- xh = h - h0 - T0(s – s0)
Calculation of incoming and outgoing exergies (2/2)
- chemical exergy: maximum work that could be produced
-
two types of changes in chemical composition to be considered :
- mixture of gases (Δs = - r Σ xi ln xi > 0)
- combustion reactions (close to the LHV or the HHV)
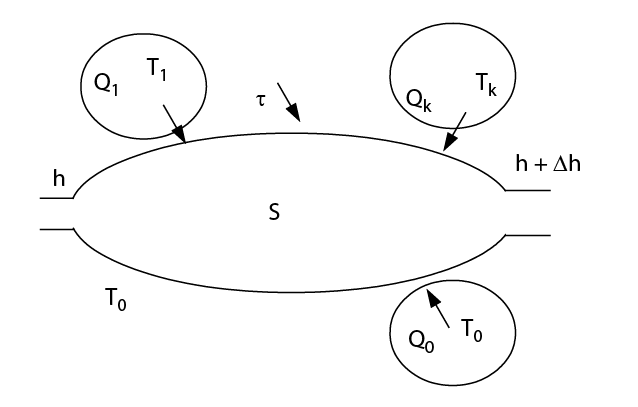
Exergy balances
- open multitherm system crossed by several fluids and involving several shaft works
Exergy balances
- exergy efficiency:
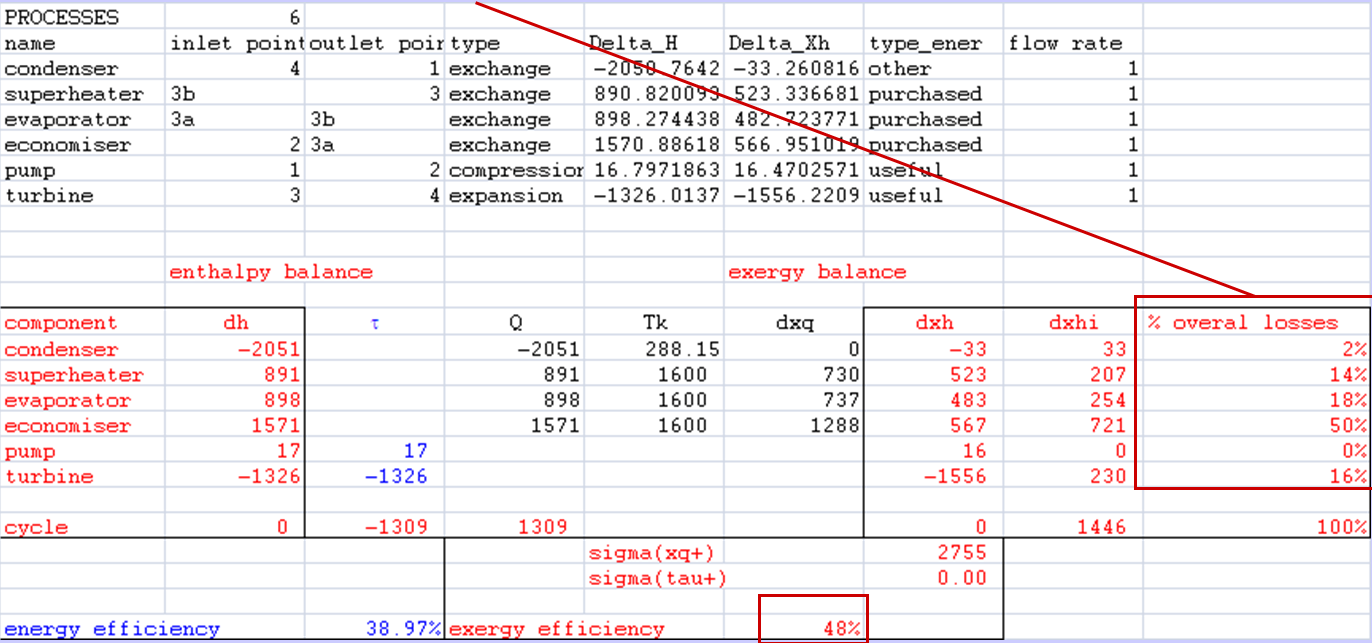
- ratio of exergy uses to exergy resources
- 1 minus (sum of exergy losses) / exergy resources
Exergy balances
- adiabatic processes: Q = 0, τ = mΔh
processes without work and with heat exchange with a source at T :
,
Presentation of the spreadsheet
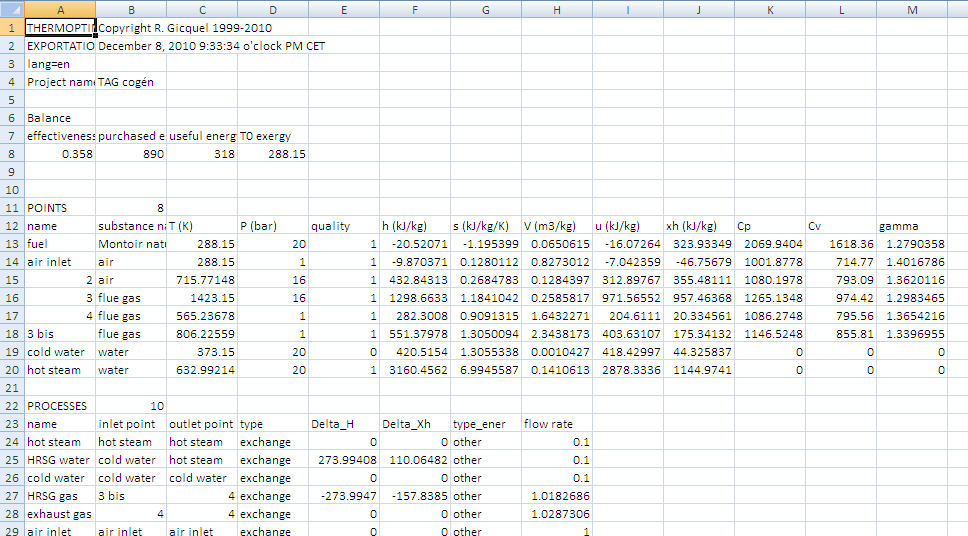
In practical terms, the exergy balance of a cycle can be established in the following manner when it is modeled in Thermoptim.
Once the model is properly set and calculated the results file can be exported (line “Export exergy calculations” of menu “Result Files”).
The first lines of this file can then be copied into the spreadsheet. You must then carefully correct by hand the part of the worksheet that makes the exergy balance, as the lines for the different processes should be constructed differently depending on whether they relate to adiabatic expansion or compression or heat exchanges with the surroundings. For the latter, you must also specify the value of the source temperature.
Finally, you should take into account the powers supplied to the system from outside. If you only have the demo version of Thermoptim you must slightly modify the spreadsheet to recalculate the exergies brought into play in the processes from the values calculated in the points screens and from the flow-rates involved, which must be copied one by one.
To facilitate the preparation of exergy balances, two documents are available from the links below:
- A methodological guide that explains how to perform the calculations, including providing some explanation of how to deal with three specific cases: taking into account the exergy of a fuel in a combustion chamber, the calculation of exergy losses in an open cycle, where the exhaust gases are released into the atmosphere, and finally how to deal with internal heat exchangers, such as in a combined cycle;
- The second document is the spreadsheet that includes a number of worksheets on Thermoptim examples.
Lecture notes on “Exergy balances” explain in detail how to proceed.
The following activities explain the structure of the spreadsheet.
Thermoptim results file
Exergy balances
- links between cells
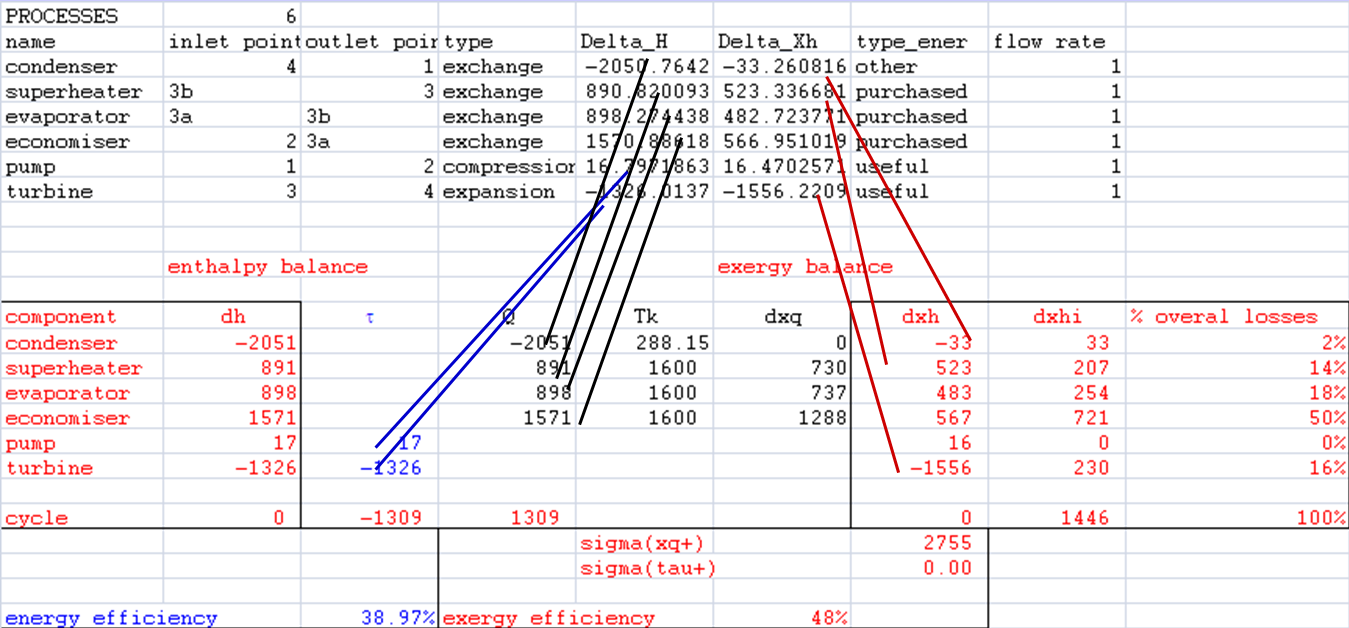
Exergy balances
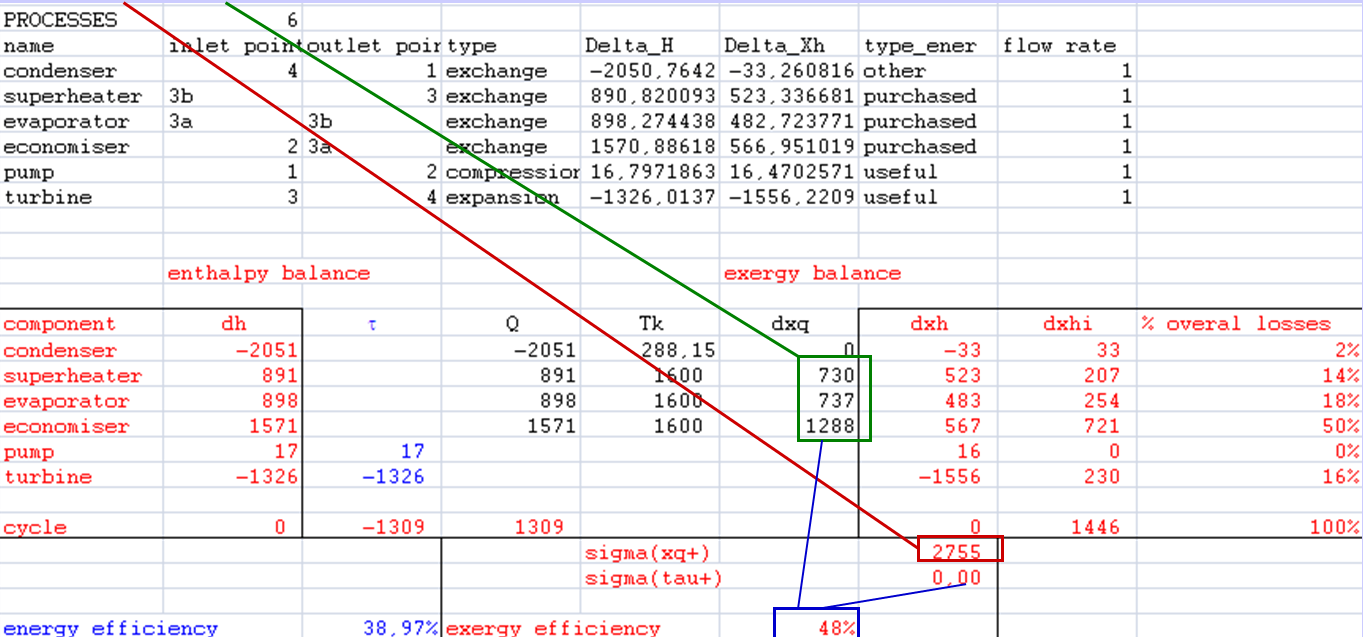
- dxhi = τ + dxq - dxh
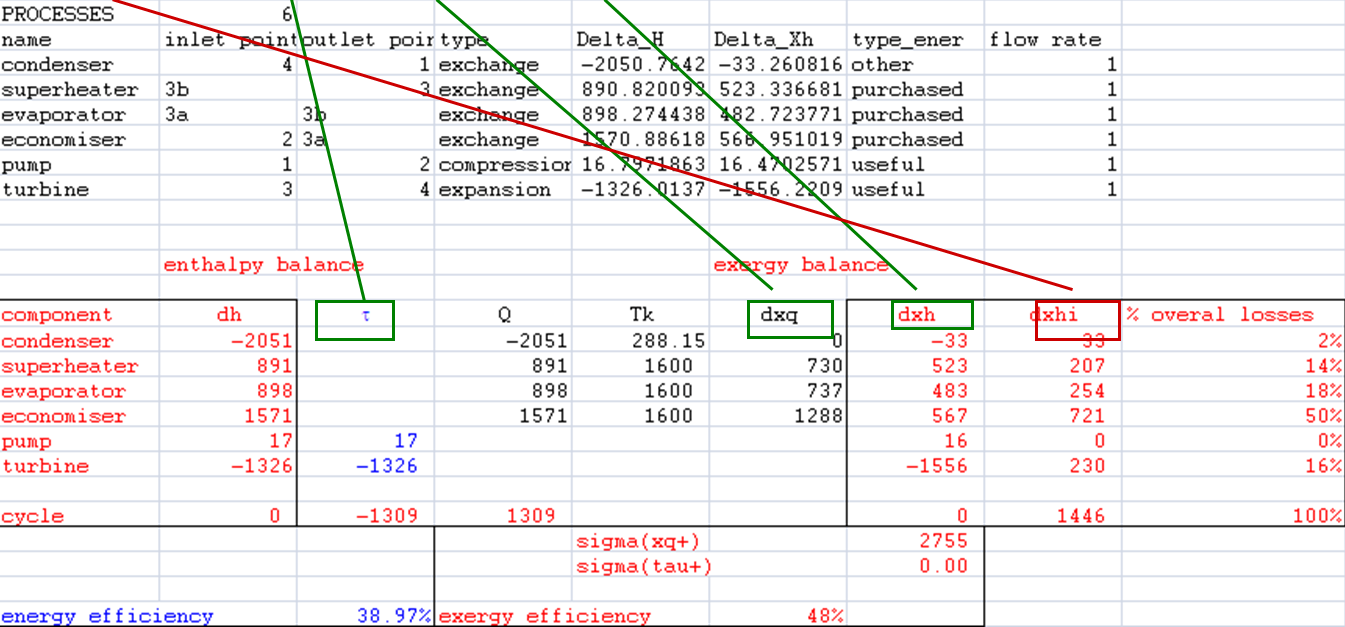
Exergy balances
- sigma(xq+) (here, sigma(tau+) = 0)
Exergy balances
- distribution of irreversibilities
Exergy balances
- Set up an exergy balance is:
- Account for exergy losses of each component
- Calculate the exergy efficiency
- Methodology for doing so from Thermoptim:
- Copy and paste the results file lines in the spreadsheet
- Edit the spreadsheet rows depending on components (refer to the methodological guide )
- Not difficult in itself, but must be done rigourously
- Many examples in the spreadsheet
Exergy balances
This session explains how to set up the exergy balance of a cycle.
It shows how to use the Thermoptim results into a spreadsheet.
We will apply this methodology to analyze quantitatively the irreversibility of various energy systems:
- A Gas Turbine (session S23En)
- Steam power plants (session S28En)
- A refrigerating machine (session S32En)
- A heat pump (session S33En)
- Combined cycle (session S41En)
- A cogeneration facility (session S47En)